|
10/11/2021 0 Comments Ni Dmg 2
Komplexe von Co, Fe, Ni, Pd und Re sind zur quantitativen. Als qualitativer Nachweis lassen sich die Komplexe unter den jeweils geeigneten Bedingungen ( pH und Temperatur) f&228 llen. Dimethylglyoxim ist ein Komplexbildner, der mit Schwermetallionen wie Ni 2+, Fe 2+, Co 2+, Cu 2+, Pt 2+, Pd 2+ und Re 7+ farbige Chelatkomplexe bildet.As you can see nickel has not been oxidized, nickel is not oxidized as the Ni2+ ion only forms a complex. Ni (DMG)2 (s) is a red precipitate. The beam was Cu K a and the scan.Thyssen, Jacob P Skare, Lizbet Lundgren, LennartNi Dmg 2 Molecular Weight Loss. Therefore, each DMG ligand has a 1- charge and the pair of DMG ligands offset the 2+ charge on the Ni ion.Download scientific diagram X-ray diffraction of Ni(DMG) 2 powder and thin AM and CR films grown on glass substrates. 0 0.0 falseCORRECT Notice that each DMG loses a single H + ion during the reaction. So if you fill up the energy levels you will get two unpaired electrons in ground state.Sensitivity and specificity of the nickel spot ( dimethylglyoxime) testIn Ni (DMG)2 the nickel is in the +2 oxidation state and to have a square planar geometry because of chelation the pairing of electrons takes place.The EN 1811, a European standard reference method developed by the European Committee for Standardization (CEN), is fine-tuned to estimate nickel release around.Sensitivity and specificity of the nickel spot ( dimethylglyoxime) test.Thyssen, Jacob P Skare, Lizbet Lundgren, Lennart Menné, Torkil Johansen, Jeanne D Maibach, Howard I Lidén, CarolaThe accuracy of the dimethylglyoxime ( DMG) nickel spot test has been questioned because of false negative and positive test reactions. Ni(DMG)2 (s) + 3 en (aq), The bright red Ni(DMG)2 precipitate is formed.The accuracy of the dimethylglyoxime ( DMG) nickel spot test has been questioned because of false negative and positive test reactions. Search results for dmg at Sigma-Aldrich.The following solutions are added to a beaker containing nickel (II) sulfate.It may serve well for screening purposes. The DMG test has a high specificity but a modest sensitivity. The sensitivity of the DMG test was 59.3% and the specificity was 97.5% based on DMG-test results and nickel release concentrations determined by the EN 1811 reference method. DMG spot testing, chemical analysis according to the EN 1811 reference method, and X-ray fluorescence spectroscopy (XRF) were performed concomitantly on 96 metallic components from earrings recently purchased in San Francisco. To evaluate the sensitivity and specificity of the DMG test. Because assessments according to EN 1811 are expensive to perform, time consuming, and may destruct the test item, it should be of great value to know the accuracy of the DMG screening test.
Ni Dmg 2 Lexe Von CoBecause assessments according to EN 1811 are expensive to perform, time consuming, and may destruct the test item, it should be of great value to know the accuracy of the DMG screening test.Nickel deposited on the skin-visualization by DMG test.Julander, Anneli Skare, Lizbet Vahter, Marie Lidén, CarolaNickel is the most common cause of contact allergy and an important risk factor for hand eczema. The limit value of the EU Nickel Directive from products intended to come into direct and prolonged skin contact. The EN 1811, a European standard reference method developed by the European Committee for Standardization (CEN), is fine-tuned to estimate nickel release around. The DMG test and acid wipe sampling for quantification were then performed. Nickel solutions at different concentrations were applied in duplicate on the hands in healthy subjects (n = 5). To develop and evaluate methods for visualization of nickel on the skin by the DMG test and hand imprints. The dimethylglyoxime ( DMG) test might be used to establish skin exposure to nickel. 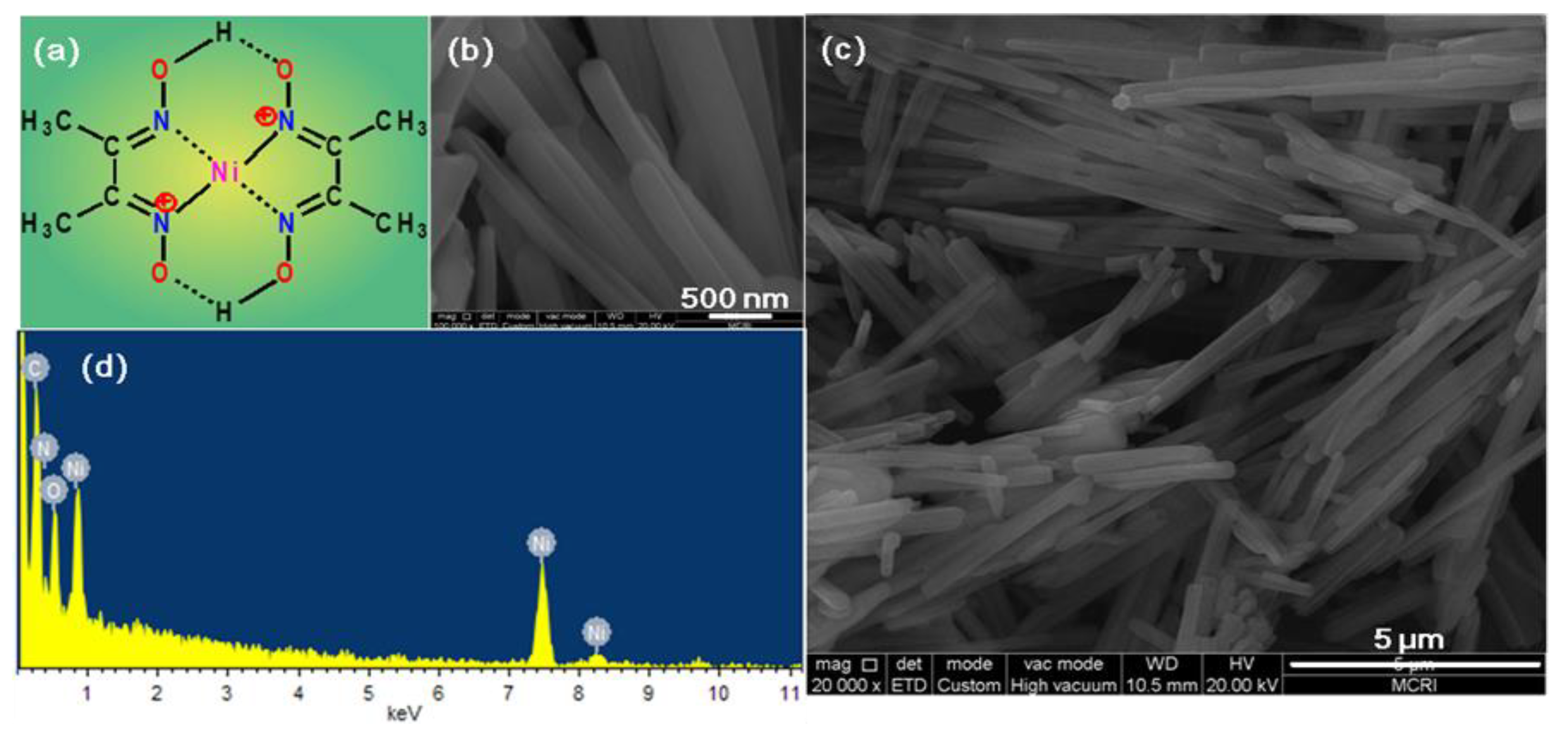
© 2011 John Wiley & Sons A/S.Optimal synthesis of a Ni(II)-dimethylglyoxime ion-imprinted polymer. DMG test-developed hand imprints may, in the future, be used for semi-quantitative or quantitative exposure assessment. The DMG test may be used as a simple and powerful tool for visualization of nickel on skin. The effects of analytical parameters such as pH, dose of DMG, concentration of nickel solution, contact time and selectivity were studied and the optimal operation parameters were found as follows: pHPZC: 7.6, CNi(II): 0.01 M, contact time: 360 min and DMG dosage: 5mM. It was found that NCP- DMG has higher capacity for removal of Ni(II). Removal of Ni(II) by modified and unmodified samples was investigated in batch procedure. The raw and modified samples were characterized by XRD, FT-IR, SEM, BET, TG-DTG and energy dispersive analysis X-ray spectroscopy (EDAX). Modification of micro and nano particles and also their Ni-exchanged forms were done by dimethylglyoxime ( DMG). This aimed to improve the performance of this ion-imprinted polymer in trapping Ni(II) ions from soil and mine tailing samples .Effective removal of Ni(II) from aqueous solutions by modification of nano particles of clinoptilolite with dimethylglyoxime.Nezamzadeh-Ejhieh, Alireza Kabiri-Samani, MehdiIn this work an Iranian natural clinoptilolite tuff was pre-treated and changed to the micro (MCP) and nano (NCP) particles by mechanical method. The negative ΔS indicates that the adsorption of nickel cations from solution occurs with lower amount ion replacement, thus chemisorptions due to complex formation are dominant process in nickel removal. The negative ΔH and ΔG indicate an exothermic and spontaneously process. Comparison of two kinetic models indicates that the adsorption kinetic can be well described by the pseudo-second-order rate equation that indicates that the rate limiting step for the process involves chemical reaction. Adsorption isotherms of Ni(II) ions could be best modelled by Langmuir equation, that indicate the monolayer sorption of Ni(II). Langmuir and Freundlich isotherm models were adopted to describe the adsorption isotherms. To identify nickel release and metal content in earrings purchased in China and Thailand. (author)Assessment of nickel release from earrings randomly purchased in China and Thailand using the dimethylglyoxime test.Hamann, Carsten R Hamann, Dathan J Hamann, Quinlan J Hamann, Curtis P Boonchai, Waranya Li, Lin-Feng Thyssen, Jacob PChina and Thailand produce large amounts of jewellery that are sold domestically and abroad. PAN- DMG material ( dimethylglyoxime in porous beads of polyacrylonitrile) was compared with Ni Resin and DMG-PAN material exhibited higher sorption capacity for nickel than Ni Resin. Quantitative separation from 100 mL of real concentrate on the column filled with 3 mL of PAN- DMG composite material was achieved. All rights reserved.59 Ni and 63 Ni separation from boric acid concentrates produced at NPPInternational Nuclear Information System (INIS)Procedure for direct separation of radionickel from boric acid concentrate was developed. Three (11.5%) of 29 DMG negative earrings contained nickel as a major component 7 (26.9%) of 29 DMG negative earrings contained nickel as a minor component. A total of 314 Chinese earrings (31.5%) and 243 Thai earrings (29.2%) were DMG test positive. Earrings were subjected to dimethylglyoxime ( DMG) tests and 26 of the DMG negative earrings were further evaluated qualitatively for major and minor metal content using X-ray fluorescence spectrometry. In China, enforcement of the 2002 Chinese National Standard GB 11887 may assist in reducing sensitization. Increased public education together with the adoption of a regulatory intervention in Thailand may be warranted. This may contribute to the high prevalence of nickel allergy in both countries. The proposed method was tested with two types of real radioactive waste (boric acid concentrate from nuclear power plant (NPP) evaporator and spent ion exchanger from NPP). Method for preparation of 59 Ni source for low energy photon spectrometry was developed using homogeneous precipitation of nickel with DMG.
0 Comments
Leave a Reply. |
AuthorDenise ArchivesCategories |
 RSS Feed
RSS Feed
